The Use of HCA and Ciclosporin in Non-Flea, Non-Food Hypersensitivity Dermatitis
HISTORY AND CLINICAL EXAM
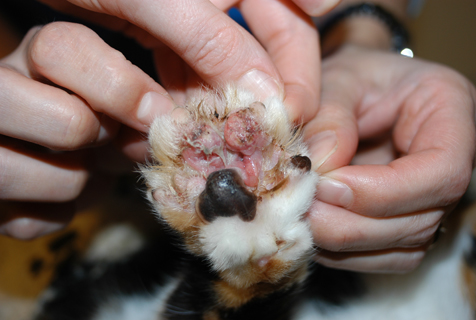
 A 4-year-old, neutered, female domestic shorthair cat presented with a two-year history of recurrent pododermatitis and self-trauma affecting all four limbs. The cat was otherwise in good health, was up to date with routine vaccinations and was fed a balanced commercial cat food. There was no history of travel and the cat predominantly lived indoors with occasional garden access.
A 4-year-old, neutered, female domestic shorthair cat presented with a two-year history of recurrent pododermatitis and self-trauma affecting all four limbs. The cat was otherwise in good health, was up to date with routine vaccinations and was fed a balanced commercial cat food. There was no history of travel and the cat predominantly lived indoors with occasional garden access.
Repeated treatment with depot methylprednisolone acetate 4mg/kg and cefovecin 8mg/kg had failed to fully control the cat’s clinical signs.
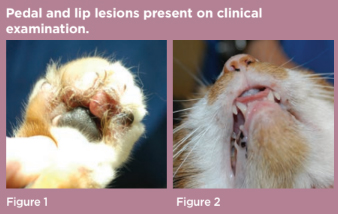
General physical examination was unremarkable. Dermatological examination revealed a well-demarcated, alopecic, erythematous and ulcerated plaque present on the palmar aspect of the right thoracic limb between digits two and four (figure 1). The surrounding pedal skin was moist and erythematous. There were also minor erosions affecting the upper lip adjacent to the canine teeth (figure 2).
DIFFERENTIALS
-
Differential diagnoses for the pedal lesion include:
-
Eosinophilic plaque (EP),
-
Neoplasia (squamous cell carcinoma, lymphoma, mast cell tumour),
-
Cutaneous viral disease (feline cowpox, herpes virus, calici virus),
-
Mycobacterial infection,
-
Deep fungal infection,
-
Bacterial folliculitis and furunculosis,
-
Foreign body reaction,
-
Sterile nodular disease.
-
-
Differential diagnoses for the lip lesion include:
-
Mild eosinophilic (indolent) ulcer
-
Trauma.
-
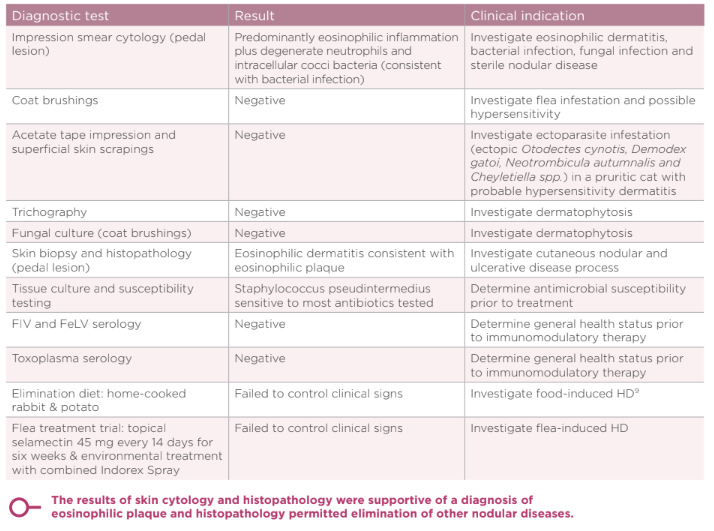
Considering the age of the cat, the clinical appearance of the pedal lesion and the presence of pruritus, EP was suspected. As eosinophilic dermatitides are seen commonly with allergic skin disease and the cat’s clinical signs fulfilled 5/8 of Favrot’s criteria for cats presenting with pruritus, a provisional diagnosis of feline hypersensitivity dermatitis (HD) was made.1,2,3
DIAGNOSTICS
TREATMENT
 The staphylococcal bacterial infection was suspected to be secondary to inflammation associated with allergic skin disease; this was treated with 8 mg/kg cefovecin for four weeks. The severity of the cat’s EP warranted symptomatic therapy prior to the completion of diagnostic trials and therefore dexamethasone was administered at 0.08 mg/kg PO SID 7 days then EOD 7 days then twice weekly for 2 weeks. This off license treatment was used as the owner was unable to administer tablets and it offered improved dosing flexibility with short-term control of the clinical disease, compared to depot steroid formulations and other licensed immunomodulatory drugs. Extensive use of this potent glucocorticoid was avoided due to the risk of adverse effects. The cat’s lesions resolved, but despite ongoing food and ectoparasite control trials, sterile eosinophilic inflammation recurred when treatment was discontinued and a diagnosis of non-flea, non-food hypersensitivity dermatitis (HD) was therefore made.4
The staphylococcal bacterial infection was suspected to be secondary to inflammation associated with allergic skin disease; this was treated with 8 mg/kg cefovecin for four weeks. The severity of the cat’s EP warranted symptomatic therapy prior to the completion of diagnostic trials and therefore dexamethasone was administered at 0.08 mg/kg PO SID 7 days then EOD 7 days then twice weekly for 2 weeks. This off license treatment was used as the owner was unable to administer tablets and it offered improved dosing flexibility with short-term control of the clinical disease, compared to depot steroid formulations and other licensed immunomodulatory drugs. Extensive use of this potent glucocorticoid was avoided due to the risk of adverse effects. The cat’s lesions resolved, but despite ongoing food and ectoparasite control trials, sterile eosinophilic inflammation recurred when treatment was discontinued and a diagnosis of non-flea, non-food hypersensitivity dermatitis (HD) was therefore made.4
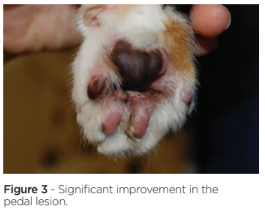
Long-term treatment with ciclosporin 7 mg/kg PO SID was commenced. Ciclosporin at this dose has been shown to be a safe and effective treatment for feline non-flea, non-food HD, including cats presenting with EP.5 The cat’s clinical signs improved on this treatment but the owner struggled with administration and the pedal lesion did not fully resolve. Concurrent topical HCA spray was therefore introduced. Two sprays were applied to the affected skin SID 7 days (figure 3) then reduced to alternate day and twice weekly treatment for maintenance therapy.

DISCUSSION
Hydrocortisone aceponate (HCA) is licensed for the treatment of canine atopic dermatitis as a 0.0584% spray.6 Daily or alternate day application has been shown to be efficacious in the treatment of cats with EP in one small study.7 Additionally, proactive therapy on two consecutive days each week has been shown to be effective and well-tolerated in the long-term management of canine atopic dermatitis.8 Potential ectoparasite-associated flares in disease were managed via continuation of topical selamectin every four weeks and twice-yearly treatment of the environment with combined permethrin / pyriproxyfen / piperonyl butoxide.

1. Bloom, P.B. (2006). Canine and feline eosinophilic skin diseases, Veterinary Clinics of North America Small Animal Practice, 36(1), p141-60.
2. Taglinger, K., Day, M.J. & Foster, A.P. (2007). Characterization of inflammatory cell infiltration in feline allergic skin disease, Journal of Comparative Pathology, 137(4), p211-23.
3. Favrot, C., Steffan, J., Seewald, W., Hobi, S., Linek, M., Marignac, G., Olivry, T., Beco, L., Nett, C., Fontaine, J. & Roosje, P. (2012). Establishment of diagnostic criteria for feline non flea-induced hypersensitivity dermatitis, Veterinary Dermatology, 23(1), p45–50.
4. Favrot, C. (2013). Feline non-flea induced hypersensitivity dermatitis clinical features, diagnosis and treatment, Journal of Feline Medicine and Surgery, 15(9), p778–84.
5. Roberts, E.S. et al (2016) ‘Confirmatory field study for the evaluation of ciclosporin at a target dose of 7.0 mg/kg (3.2 mg/lb) in the control of feline hypersensitivity dermatitides’. Journal of Feline Medicine and Surgery, 18(11), p889-97.
6. Nuttall, T., Mueller, R., Bensignor, E., Verde, M., Noli, C., Schmidt, V. & Rème, C. (2009) Efficacy of a 0.0584% hydrocortisone aceponate spray in the management of canine atopic dermatitis: a randomised, double blind, placebo-controlled trial, Veterinary Dermatology, 20(3), p191-8.
7. Schmidt, V., Buckley, L.M., McEwan, N.A., Reme, C.A. & Nuttall, T.J. (2012). Efficacy of a 0.0584% hydrocortisone aceponate spray in presumed feline allergic dermatitis: an open label pilot study, Veterinary Dermatology, 23(1), p11-6.
8. Lourenço, A.M., Schmidt, V., Sao Braz, B., Nobrega, D., Nunes, T., Duarte‐Correia, J.H., Matias, D., Maruhashi, E., Reme, C.A. & Nuttall, T. (2016). Efficacy of proactive long-term maintenance therapy of canine atopic dermatitis with 0.0584% hydrocortisone aceponate spray: a double-blind placebo controlled pilot study, Veterinary Dermatology, 27(2), p88-92.
9. Bryan, J. & Frank, L.A. (2010). Food allergy in the cat: a diagnosis by elimination, Journal of Feline Medicine and Surgery, 12(11), p861-6.

